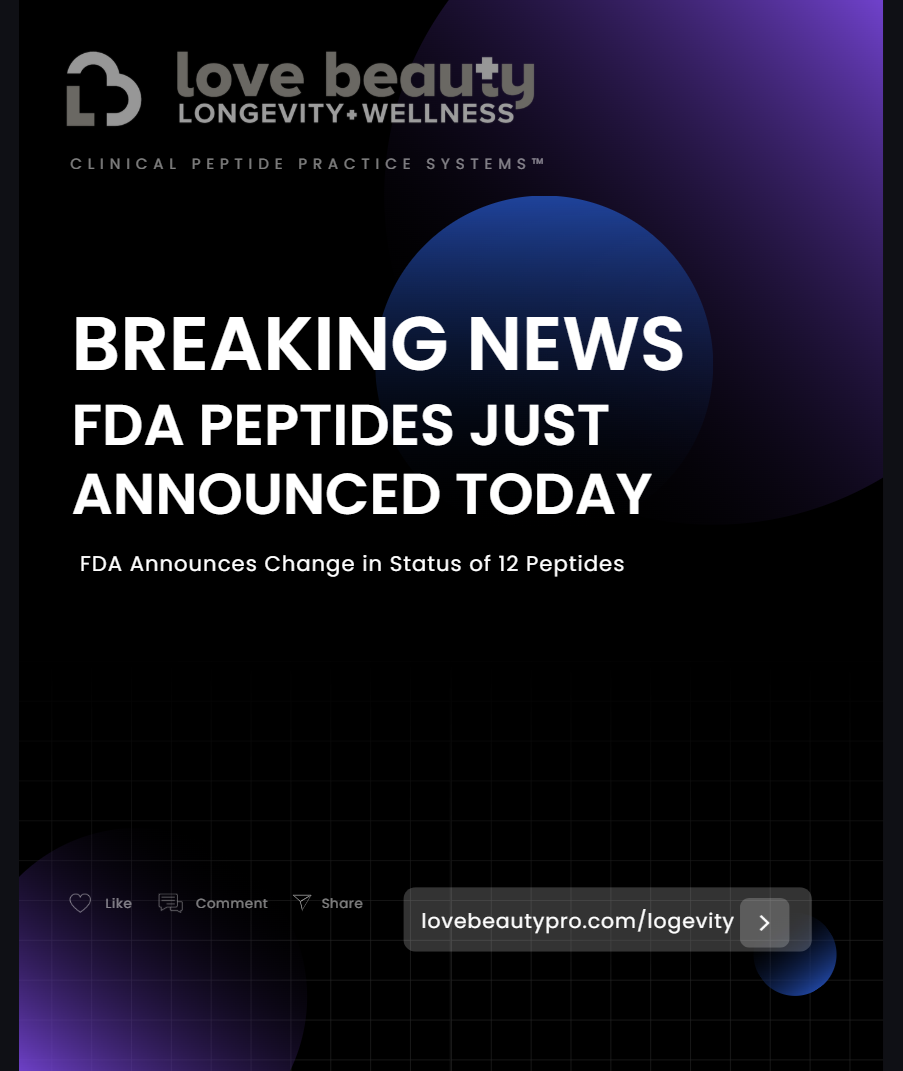
BREAKING NEWS: FDA PEPTIDES JUST ANNOUNCED TODAY – Change in Status of 12 Peptides
The FDA reported today that it will soon remove the following bulk drug substances from Category 2:
BPC-157
LL-37
DiHexa
DSIP
Epitalon
*GHK-Cu (injectable only)
KPV
PEG-MGF
Melanotan II
MOTs-C
Semax
TB-500.
Originally nominated for Category 2 (Bulk Substances that Raise Significant Safety Concerns), the withdrawal of these nominations seems to indicate the FDA may be reconsidering the validity of distinct safety concerns.
Removal from Category 2 also lessens restrictions on US 503(A) compounding pharmacies’ ability to produce these peptides. Today’s FDA announcement also suggested that its upcoming meeting in July 2026 will examine the possibility of giving these peptides full approval.
*Non-injectibile GHK-Cu has also been singled out for further consideration by February 2027.
This news is exciting for patients of US Medical Providers and Practices who increasingly seek longevity wellness and peptide therapy by trained providers.
—————————————————————————————————————————————————————————————————
FDA Updates 503A Bulk Drug Substances Framework and Advances Review of Peptide Compounds
Personalized Health Association Provides Industry Perspective as Federal Review of Peptide Compounds Enters Formal Evaluation Phase
WASHINGTON DC, DC, UNITED STATES, April 15, 2026 /EINPresswire.com/ — The Personalized Health Association (PHA) today responded to the latest developments from the U.S. Food and Drug Administration regarding bulk drug substances nominated for compounding under Section 503A of the Federal Food, Drug, and Cosmetic Act, including category updates and the scheduling of forthcoming Pharmacy Compounding Advisory Committee (PCAC) hearings.
The Agency’s actions reflect continued progression from interim categorization toward a formalized evaluation pathway for multiple peptide compounds currently utilized in physician-directed care.
Regulatory Transition and Peptide Review Pathway
FDA’s update includes the removal of several peptide substances from interim categories, alongside notice of upcoming PCAC review for potential inclusion on the 503A bulks list.
These include peptides such as:BPC-157, Semax, TB-500 (Thymosin Beta-4 fragment), Epitalon, MOTs-C, KPV, and Dihexa, among others.
Additional compounds widely cited in ongoing policy discussions and scientific literature include:GHK-Cu, DSIP (Delta Sleep-Inducing Peptide), Melanotan II, PEG-MGF, LL-37 (Cathelicidin), and related peptide analogs currently under regulatory consideration.
The Agency has indicated that these substances will be subject to formal evaluation beginning in July 2026, with additional reviews extending into 2027.
While public discussion has accelerated around peptide access, formal regulatory determination remains under FDA review through the PCAC process.
Recent federal signals, including the scheduling of PCAC review and broader policy discussions regarding peptide access indicate that regulatory agencies are actively evaluating near-term pathways to expand patient access during the interim review period.
Under a framework of enforcement discretion, and consistent with physician oversight and 503A compounding standards, these compounds may be eligible for prescribing and compounding within a regulated, patient-specific context ahead of final PCAC determination.
This evolving framework reflects a growing alignment between patient demand, clinical use, and regulatory pathways, offering the potential for near-term access within a structured and medically supervised environment.
Strategic Leadership and Regulatory Engagement
Lee Rosebush, Lead Strategist for PHA, stated:
“We are now at a critical stage in the regulatory process where years of advocacy are transitioning into formal federal review. Moving these substances into the PCAC framework reflects that progression—establishing a structured pathway for evaluation grounded in safety, clinical use, and scientific data.
This moment marks the point where stakeholder engagement, policy work, and clinical reality converge into a process capable of delivering long-term regulatory clarity.”
Safety, Access, and System Integrity
Jimmy St. Louis, President of PHA, added:
“This transition is fundamentally about safety. Establishing a structured regulatory process creates a pathway for these therapies to move out of gray-market channels and into regulated, quality-controlled environments.
As clearer frameworks emerge, we are seeing a pathway for patients to transition away from unregulated sourcing and back into physician-guided care. That shift has the potential to significantly improve safety while restoring appropriate access.”
Founder Perspective
Bill Moses, Founder and Chairman of PHA, said:
“We are entering a transition from regulatory ambiguity to structured evaluation. The priority now is ensuring that patient–provider decision-making is preserved within a compliant, medically supervised framework.
If properly executed, this pathway allows innovation to advance while strengthening safety, transparency, and long-term regulatory integrity.”
Next Phase: PCAC Review
FDA has scheduled multiple peptide-related substances for review by the Pharmacy Compounding Advisory Committee beginning in July 2026, with additional sessions anticipated into 2027.
PHA will continue to engage constructively throughout this process, supporting:
• Evidence-based evaluation
• High-quality manufacturing and sourcing standards
• Clear regulatory pathways within existing statutory frameworks
• Integration of clinical and real-world safety data
About the Personalized Health Association (PHA)
The Personalized Health Association (PHA) is a national 501(c)(4) advocacy organization advancing safe, responsible access to regenerative and precision medicine. PHA leads federal and state efforts to modernize policies governing adult stem cell, peptide, and regenerative therapies, including advocating for updates to 21 C.F.R. §1271.10(a) to establish a clear, cGMP-based pathway for certain regenerative products to be regulated as 361 HCT/Ps.
PHA represents healthcare providers, accredited regenerative-medicine facilities, compounding pharmacies, peptide-therapy providers, telemedicine platforms, and other healthcare stakeholders committed to high standards of safety, compliance, and evidence-based care. Through coordinated policy leadership, PHA champions physician autonomy and patient access to individualized therapies. Learn more at www.personalizedhealthassociation.org
Candace McDonald
Personalized Health Association
+1 310-699-6311
email us here
Visit us on social media:
LinkedIn
Legal Disclaimer:
EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
Source Credit: https://www.einpresswire.com/article/905946942/fda-updates-503a-bulk-drug-substances-framework-and-advances-review-of-peptide-compounds
———————————————————————————————————————————————————-
MORE TO COME
What impact will FDA’s peptide review have on drug compounding?
How might PCAC hearings influence peptide therapy regulations?
What are the safety implications of regulating peptide compounds?